We recognize that accurate detection methods are essential for studying these cellular changes. Western blotting has emerged as the gold standard technique for analyzing protein modifications in laboratory settings. This method provides researchers with reliable, quantitative results that drive scientific discovery forward.
The technique offers unmatched precision for identifying specific protein states within complex biological samples. You can achieve consistent, reproducible results when proper protocols are followed. This analytical approach has revolutionized our understanding of signal transduction pathways and cellular communication networks.
Modern research demands robust detection methods that deliver accurate data. The combination of immunodetection principles with electrophoretic separation creates a powerful analytical tool for protein research applications.
Key Takeaways
- Protein phosphorylation serves as a crucial regulatory mechanism controlling cellular processes like metabolism and apoptosis
- Western blotting provides the gold standard method for detecting and analyzing protein modifications
- This technique offers precise, quantitative results essential for molecular biology research
- Proper protocol implementation ensures consistent and reproducible analytical outcomes
- The method combines immunodetection with electrophoretic separation for enhanced specificity
- Applications span across multiple research disciplines requiring protein analysis capabilities
Understanding Phosphorylated Proteins and Their Importance
Phosphoprotein detection methods have become essential tools for modern biological research. These techniques allow scientists to study one of the most fundamental regulatory mechanisms in living cells. We recognize that understanding protein phosphorylation is crucial for advancing research in multiple scientific disciplines.
The significance of phosphorylated proteins extends far beyond basic cellular functions. You will discover how these modified proteins control everything from cell division to disease progression. This knowledge forms the foundation for developing targeted therapies and diagnostic tools.
What are Phosphorylated Proteins?
Phosphorylated proteins are molecules that have undergone a specific chemical modification. Protein kinases catalyze the covalent addition of phosphate groups to serine, threonine, and tyrosine amino acid residues. This process represents one of the most common post-translational modifications in cells.
The phosphorylation process is reversible. Phosphatase enzymes remove these phosphate groups through dephosphorylation reactions. This dynamic balance between phosphorylation and dephosphorylation creates a sophisticated regulatory system.
These modifications occur rapidly in response to cellular signals. The addition or removal of phosphate groups can dramatically alter protein function. Understanding this mechanism is essential for researchers working with phosphoprotein detection methods.
Role of Phosphorylation in Cellular Signaling
Protein phosphorylation serves as a molecular switch in cellular communication. Signal transduction cascades rely heavily on phosphorylation events to transmit information throughout the cell. These cascades control critical processes including cell growth, differentiation, and apoptosis.
Phosphorylation affects protein activity in multiple ways. It can activate or inhibit enzyme function, alter protein-protein interactions, and change subcellular localization. These modifications enable cells to respond quickly to environmental changes and internal signals.
The specificity of kinase-substrate interactions ensures precise cellular responses. Different kinases target specific amino acid sequences, creating distinct signaling pathways. This specificity allows cells to maintain complex regulatory networks without unwanted cross-reactions.
Relevance in Disease Research
Aberrant phosphorylation patterns contribute to numerous pathological conditions. Cancer research heavily relies on studying dysregulated kinase activity and abnormal phosphorylation events. These alterations often serve as both disease markers and therapeutic targets.
Neurological disorders frequently involve disrupted phosphorylation cascades. Alzheimer’s disease, Parkinson’s disease, and other neurodegenerative conditions show characteristic phosphorylation abnormalities. Researchers use advanced detection methods to identify these disease-specific patterns.
| Disease Category | Key Phosphorylation Targets | Research Applications | Clinical Relevance |
|---|---|---|---|
| Cancer | p53, Rb, EGFR | Biomarker discovery | Targeted therapy development |
| Neurological Disorders | Tau, α-synuclein, APP | Disease mechanism studies | Diagnostic marker identification |
| Metabolic Diseases | Insulin receptor, AMPK | Pathway analysis | Drug efficacy testing |
| Cardiovascular Disease | Troponin, PKA substrates | Signal pathway mapping | Risk assessment tools |
Metabolic diseases also demonstrate the importance of phosphorylation research. Diabetes, obesity, and related conditions involve disrupted kinase signaling pathways. Understanding these mechanisms helps researchers develop more effective treatments and prevention strategies.
The pharmaceutical industry increasingly focuses on kinase inhibitors as therapeutic agents. These drugs target specific phosphorylation events involved in disease processes. Successful development requires sophisticated detection methods to monitor drug effects and optimize treatment protocols.
The Basics of Western Blotting Technique
Understanding western blot fundamentals enables accurate phosphorylation site analysis across diverse research applications. This immunoblotting technique combines protein separation with specific antibody detection to identify target proteins in complex biological samples. We provide comprehensive guidance on methodology, equipment requirements, and procedural considerations essential for successful phosphoprotein detection.
Overview of the Western Blot Process
The western blot protocol follows five sequential steps that ensure reliable protein identification. Gel electrophoresis separates proteins by molecular weight using polyacrylamide gels. This separation creates distinct protein bands based on size differences.
Transfer protocols move separated proteins from gels onto nitrocellulose or PVDF membranes. The transfer process preserves protein positioning while enabling antibody access. Blocking procedures prevent non-specific antibody binding by saturating membrane surfaces with inert proteins.
Primary antibody incubation targets specific protein epitopes with high specificity. Secondary antibody systems amplify detection signals through enzyme conjugation or fluorescent labeling. Detection substrates generate visible signals that correspond to target protein locations.
Key Equipment and Reagents Needed
Successful western blotting requires specialized equipment and high-quality reagents. Criterion TGX Stain-Free Gels provide consistent protein separation with built-in visualization capabilities. These precast gels eliminate preparation variability while ensuring reproducible results.
| Equipment Category | Essential Items | Key Specifications | Application Purpose |
|---|---|---|---|
| Electrophoresis System | Power supply, gel tank, electrodes | 100-300V capacity, temperature control | Protein separation by size |
| Transfer Apparatus | Transfer tank, cooling system, membranes | Constant current, 4°C operation | Protein immobilization |
| Detection System | Imaging equipment, substrates | Chemiluminescent or fluorescent | Signal visualization |
| Reagents | Clarity Western ECL Substrate Kit | Enhanced sensitivity, low background | Signal amplification |
Clarity Western ECL Substrate Kit delivers enhanced chemiluminescent detection with minimal background interference. Specialized blocking buffers prevent non-specific binding while maintaining antibody accessibility to target epitopes.
Advantages of Western Blot for Protein Detection
Western blotting offers unique advantages for phosphorylation site analysis compared to alternative detection methods. Specificity remains the primary benefit, as antibodies recognize precise protein modifications with minimal cross-reactivity. This specificity enables researchers to distinguish between phosphorylated and non-phosphorylated protein forms.
Quantitative capabilities allow accurate measurement of protein expression levels and modification states. Signal intensity correlates directly with protein abundance, enabling comparative analysis across experimental conditions. Semi-quantitative results provide reliable data for statistical analysis.
Sample compatibility extends across diverse biological materials including cell lysates, tissue extracts, and purified proteins. The technique accommodates various sample volumes and protein concentrations. Cost-effectiveness makes western blotting accessible for routine laboratory applications while maintaining research-grade accuracy.
Reproducibility ensures consistent results across experimental replicates and different laboratories. Standardized protocols minimize technical variation while preserving detection sensitivity. These advantages establish western blotting as the preferred method for phosphoprotein analysis in research and diagnostic applications.
Specific Techniques for Detecting Phosphorylated Proteins
The detection of phosphorylated protein states presents unique challenges that require targeted experimental approaches and validation methods. Immunoblotting phosphoproteins demands specialized techniques that go beyond standard protein detection protocols. We provide comprehensive guidance on methodological considerations that ensure accurate and reproducible results in phosphorylation studies.
Successful phosphoprotein detection relies on three critical components: antibody specificity, signal enhancement strategies, and rigorous experimental controls. These elements work together to create a robust analytical framework for studying protein phosphorylation events.
Phospho-specific Antibodies
Phospho-specific antibodies represent the cornerstone of successful immunoblotting phosphoproteins experiments. These specialized reagents recognize only the phosphorylated form of target proteins, providing exceptional specificity for modified protein states.
When selecting phospho-specific antibodies, you must consider several key factors. Cross-reactivity testing ensures the antibody does not bind to non-phosphorylated proteins or unrelated phosphorylated targets. Validation data should demonstrate clear discrimination between phosphorylated and non-phosphorylated protein forms.
We recommend evaluating antibody performance using positive and negative control samples. Positive controls include cells treated with kinase activators or phosphatase inhibitors. Negative controls feature samples treated with specific phosphatases that remove the target phosphorylation.
Enhancing Sensitivity in Detection
Signal enhancement techniques significantly improve the detection of low-abundance phosphoproteins. Protein loading optimization ensures adequate sample concentration while avoiding gel overloading that can compromise resolution.
Substrate selection plays a crucial role in sensitivity enhancement. Chemiluminescent substrates provide superior sensitivity compared to colorimetric alternatives. Enhanced chemiluminescent substrates can detect femtogram quantities of target proteins.
Signal amplification methods include secondary antibody systems and biotin-streptavidin complexes. These approaches multiply the detection signal without increasing background noise. Proper incubation times and temperatures maximize antibody binding efficiency while maintaining specificity.
Importance of Control Experiments
Control experiments validate the specificity of phosphoprotein detection and confirm that observed signals represent genuine phosphorylation events. Phosphatase treatment controls serve as the gold standard for verifying antibody specificity.
Lambda protein phosphatase treatment removes phosphate groups from serine, threonine, and tyrosine residues. Samples treated with phosphatase should show complete loss of signal when probed with phospho-specific antibodies. Persistent signals indicate non-specific antibody binding.
Additional control experiments include peptide competition assays using phosphorylated and non-phosphorylated peptides corresponding to the antibody target sequence. These controls further validate antibody specificity and help optimize experimental conditions for reliable immunoblotting phosphoproteins detection.
Application Scenarios for Western Blot in Research
Across the spectrum of life sciences research, western blot analysis provides unparalleled insights into protein kinase activity and cellular signaling mechanisms. This versatile technique enables researchers to investigate phosphorylation dynamics under diverse experimental conditions. We observe significant applications spanning cancer biology, neurological studies, and pharmaceutical development.
Time-course experiments represent a critical application where researchers track transient phosphorylation events. These studies reveal how cellular stimulation triggers specific kinase cascades. You can monitor protein modifications occurring within minutes or hours of treatment.
The phosphorylated protein has been detected
Cancer Studies and Biomarker Identification
Cancer research heavily relies on western blot analysis to understand oncogenic signaling pathways. Researchers examine how tumor cells exhibit altered phosphorylation patterns compared to healthy tissue. Biomarker identification becomes possible through systematic analysis of phosphoprotein expression.
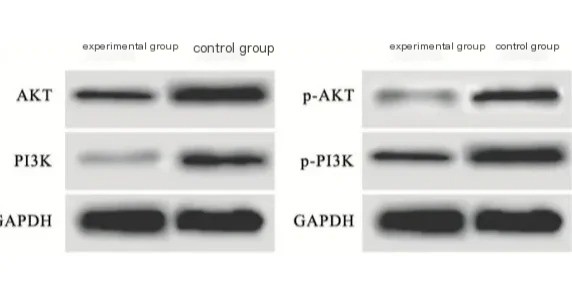
Key oncogenic pathways like PI3K/AKT and MAPK show distinct phosphorylation signatures in different cancer types. We can detect these modifications to classify tumor subtypes and predict treatment responses. Drug resistance mechanisms often involve changes in kinase activity that western blot readily identifies.
Clinical applications include monitoring patient samples for therapeutic targets. Phosphorylated proteins serve as indicators of pathway activation status. This information guides personalized treatment strategies.
Neurological Disorders and Signal Pathways
Neurological research utilizes phosphoprotein analysis to investigate synaptic signaling and neurodegeneration mechanisms. Protein kinase activity plays crucial roles in memory formation, synaptic plasticity, and neuronal survival. Alzheimer’s disease research focuses on tau protein phosphorylation patterns.
Parkinson’s disease studies examine alpha-synuclein modifications and their impact on neuronal function. Researchers track how disease progression correlates with specific phosphorylation events. These findings reveal potential therapeutic targets for intervention.
Synaptic signaling research benefits from western blot’s ability to detect activity-dependent phosphorylation. You can measure how learning and memory processes alter protein modifications in brain tissue.
Drug Development and Pharmacodynamics
Pharmaceutical development relies on western blot analysis to understand drug mechanisms and efficacy. Researchers monitor how compounds affect target protein phosphorylation in dose-response studies. This approach validates drug targets and optimizes dosing regimens.
Mechanism-of-action investigations use phosphorylation analysis to confirm drug specificity. We track downstream signaling changes to understand therapeutic effects. Off-target effects become apparent through unexpected phosphorylation patterns.
Pharmacodynamic studies employ time-course experiments to determine optimal treatment timing. Researchers measure how long drug effects persist through phosphoprotein monitoring. This data supports clinical trial design and regulatory submissions.
Comparing Western Blot with Other Protein Analysis Methods
Modern protein analysis offers several approaches for detecting post-translational modifications, each with distinct advantages. We understand that selecting the optimal method depends on your research objectives and experimental constraints. The choice between techniques significantly impacts data quality and research outcomes.
Different analytical methods provide varying levels of sensitivity, specificity, and throughput capabilities. Your decision should consider factors such as sample availability, required quantification precision, and budget limitations. We recommend evaluating multiple criteria before committing to a specific approach.
ELISA vs. Western Blot for Phosphorylation Detection
ELISA-based assays offer high-throughput screening capabilities for phosphorylated proteins in large sample sets. These methods provide quantitative results with excellent reproducibility across multiple experiments. You can process hundreds of samples simultaneously using automated platforms.
Western blotting delivers superior specificity for post-translational modifications through protein size verification. This technique allows you to distinguish between different protein isoforms and confirm molecular weight accuracy. The visual confirmation of band patterns provides additional validation that ELISA cannot offer.
Sensitivity considerations favor ELISA for detecting low-abundance phosphoproteins in complex samples. However, western blot excels when you need to analyze multiple phosphorylation sites on the same protein. The choice depends on whether you prioritize throughput or detailed protein characterization.
Mass Spectrometry for Phosphoproteomics
Mass spectrometry provides comprehensive identification of post-translational modifications across entire proteomes. This technique offers unparalleled capability for discovering novel phosphorylation sites and quantifying modification stoichiometry. You gain access to site-specific information that other methods cannot provide.
The combination of liquid chromatography with tandem mass spectrometry enables precise localization of phosphorylation events. We recognize that this approach generates extensive datasets requiring specialized bioinformatics analysis. The technique excels in discovery-phase research where comprehensive mapping is essential.
Cost considerations make mass spectrometry less accessible for routine validation studies. Western blotting remains more practical for targeted analysis of known post-translational modifications. The complementary nature of these techniques often requires integrated experimental approaches.
When to Use Western Blot over Other Methods
Western blotting proves optimal when you need visual confirmation of protein modifications with molecular weight verification. This method excels in validation studies following initial discovery through other techniques. You should choose western blot when sample throughput requirements are moderate.
Budget constraints often favor western blotting over mass spectrometry for targeted phosphorylation analysis. The technique requires standard laboratory equipment available in most research facilities. We recommend western blot when you need reliable, reproducible results for publication-quality data.
| Method | Sensitivity | Specificity | Throughput | Cost |
|---|---|---|---|---|
| Western Blot | High | Excellent | Moderate | Low |
| ELISA | Very High | Good | Very High | Moderate |
| Mass Spectrometry | Excellent | Excellent | High | High |
Time constraints influence method selection significantly in research environments. Western blotting provides results within hours, while mass spectrometry may require days for complete analysis. Your experimental timeline should guide the decision-making process.
The nature of your research question determines the most appropriate analytical approach. Discovery studies benefit from mass spectrometry’s comprehensive coverage, while validation experiments favor western blot’s targeted specificity. We suggest considering the entire experimental workflow when selecting methods for post-translational modifications analysis.
Requirements for Successful Western Blot Experiments
Successful western blot detection of phosphorylated proteins depends on precise technical requirements and standardized procedures. We recommend following established protocols that address critical experimental parameters from sample collection through final detection. You must maintain strict control over each step to ensure reproducible and reliable results.
The integrity of your experimental design directly impacts the quality of your data. Proper planning and execution of these requirements will maximize your chances of detecting target phosphoproteins while minimizing background interference.
Sample Preparation and Protein Extraction
Effective sample preparation forms the foundation of successful SDS-PAGE phosphoproteins analysis. You must include phosphatase inhibitors in your lysis buffer to prevent dephosphorylation during extraction. We recommend using a cocktail of both serine/threonine and tyrosine phosphatase inhibitors for comprehensive protection.
Your lysis buffer composition should contain protease inhibitors alongside phosphatase inhibitors. Immediate processing of samples after collection prevents degradation of phosphorylation states. Store samples at -80°C if immediate analysis is not possible.
Consider these essential components for optimal protein extraction:
- RIPA buffer or similar detergent-based lysis solution
- Phosphatase inhibitor cocktail (1:100 dilution)
- Protease inhibitor cocktail (1:100 dilution)
- DTT or β-mercaptoethanol for reducing conditions
Gel Electrophoresis Considerations
Gel selection significantly impacts the separation quality of your phosphorylated proteins. We suggest using gradient gels (4-20%) for optimal resolution across different molecular weights. Bis-Tris gels provide superior band sharpness compared to traditional Tris-glycine systems.
Running conditions require careful optimization for phosphoprotein detection. Lower voltage settings (100-120V) prevent band distortion and maintain protein integrity. You should run gels at 4°C when possible to minimize heat-induced artifacts.
Transfer efficiency directly affects your final results. Use PVDF membranes for better protein binding capacity compared to nitrocellulose. Optimize transfer time and voltage based on your target protein size to ensure complete transfer without over-heating.
Proper Blocking and Antibody Dilutions
Blocking strategy requires special consideration for phosphoprotein detection. Avoid milk-based blocking solutions due to casein phosphoproteins that can interfere with phospho-specific antibodies. We recommend using 5% BSA in TBS-T for optimal blocking performance.
Buffer selection plays a crucial role in maintaining antibody specificity. Use Tris-based buffers rather than phosphate-based systems to prevent competition with phospho-epitopes. This approach enhances signal-to-noise ratios significantly.
Antibody dilution optimization ensures maximum sensitivity while minimizing background. Start with manufacturer recommendations and perform titration experiments. Primary antibody incubation should occur overnight at 4°C for phospho-specific antibodies to achieve optimal binding.
Consider these blocking and antibody guidelines:
- Block membranes for 1-2 hours at room temperature
- Use fresh blocking solution for each experiment
- Dilute antibodies in blocking solution containing 0.02% sodium azide
- Include appropriate positive and negative controls
Troubleshooting Common Issues in Western Blotting
Identifying and resolving common issues ensures reliable protein phosphorylation assay results. We understand that technical challenges can arise at any stage of the experimental process. Systematic troubleshooting approaches help you maintain consistent data quality and experimental reproducibility.
Successful problem resolution requires understanding the root causes of each issue. You can minimize experimental setbacks by implementing diagnostic strategies and preventive measures. Our expertise in western blotting applications enables us to guide you through the most effective solutions.
Background Noise and Non-specific Binding
High background signals often stem from inappropriate blocking reagent selection. Milk-based blocking solutions contain casein phosphoproteins that can interfere with phospho-specific antibody binding. This creates false positive signals and reduces the signal-to-noise ratio in your experiments.
We recommend using bovine serum albumin (BSA) or specialized phospho-blocking reagents instead. These alternatives provide effective protein binding sites without introducing phosphorylated contaminants. You should test different blocking concentrations to optimize your specific experimental conditions.
Antibody concentration optimization reduces non-specific binding significantly. Start with manufacturer-recommended dilutions and perform titration experiments. Lower antibody concentrations often improve specificity while maintaining adequate signal strength.
Extended blocking times and additional wash steps help eliminate residual background. You can implement these modifications without compromising your experimental timeline. Consider using automated washing systems for consistent results across multiple experiments.
Troubles with Band Visualization
Weak or absent bands indicate several potential issues in your experimental setup. Insufficient protein loading represents the most common cause of poor signal detection. We suggest verifying protein concentrations using Bradford or BCA assays before gel loading.
Phosphorylation induction protocols may require optimization for your specific cell lines. Different stimulation conditions, timing, and inhibitor treatments affect phosphorylation levels dramatically. You should establish positive controls using known phosphorylation inducers.
Detection substrate selection impacts signal intensity and duration significantly. Enhanced chemiluminescent substrates provide superior sensitivity for low-abundance phosphorylated proteins. Consider upgrading your detection system if standard substrates produce insufficient signals.
Imaging parameters require careful adjustment for optimal band visualization. Exposure times, gain settings, and binning options affect signal capture quality. You can improve weak signals by extending exposure times or increasing camera sensitivity settings.
Analyzing Inconsistent Results
Experimental variability often results from inconsistent sample preparation procedures. Standardize your protein extraction protocols and include protease inhibitors consistently. Fresh inhibitor cocktails ensure maximum protein stability throughout the extraction process.
Antibody storage and handling conditions affect performance reliability over time. Store primary antibodies according to manufacturer specifications and avoid repeated freeze-thaw cycles. You should aliquot antibodies into single-use portions for critical experiments.
Gel-to-gel variations can introduce systematic errors in your results. Use pre-cast gels when possible to ensure consistent acrylamide concentrations and pH conditions. Document all experimental parameters for troubleshooting purposes and result interpretation.
Temperature fluctuations during incubation steps affect antibody binding kinetics. Maintain consistent temperatures throughout blocking, primary antibody, and secondary antibody incubations. We recommend using temperature-controlled shakers for reproducible results.
Control experiment design helps identify sources of experimental variation. Include positive controls, negative controls, and loading controls in every experiment. Systematic control implementation enables rapid problem identification and resolution strategies.
Documentation of troubleshooting steps facilitates future problem prevention. Record all modifications, observations, and outcomes for reference purposes. This approach builds institutional knowledge and improves overall experimental success rates.
Future Trends in Western Blot and Phosphorylation Research
Revolutionary advances in detection methodologies are transforming how researchers approach phosphorylation studies. The scientific community continues to push boundaries in protein analysis technologies. These innovations promise to enhance both sensitivity and specificity in phosphoprotein detection.
Modern laboratories are witnessing unprecedented developments in analytical capabilities. We see emerging technologies that address traditional limitations while opening new research possibilities. The integration of cutting-edge detection systems with established western blot protocols creates powerful analytical platforms.
Advances in Detection Technologies
Next-generation detection systems are revolutionizing phosphoprotein analysis through enhanced sensitivity and precision. Fluorescent western blotting represents a significant leap forward in multiplexing capabilities. This technology enables simultaneous detection of total and phosphorylated proteins on the same membrane.
The development of ultra-sensitive substrates has dramatically improved signal-to-noise ratios. These substrates allow detection of low-abundance phosphoproteins that were previously undetectable. Researchers can now identify subtle phosphorylation changes in cellular signaling pathways.
Automated imaging systems are streamlining data acquisition and analysis processes. These systems reduce human error while increasing throughput capacity. Digital imaging platforms provide quantitative measurements with unprecedented accuracy and reproducibility.
Enhanced antibody specificity for phosphorylation sites continues to drive technological progress. Manufacturers are developing highly specific antibodies that recognize unique phosphorylation patterns. This advancement reduces cross-reactivity and improves experimental reliability.
Integration of Western Blot with Other Techniques
The convergence of western blot technology with complementary analytical methods creates comprehensive research platforms. Mass spectrometry integration provides molecular weight confirmation and phosphorylation site mapping. This combination offers both qualitative and quantitative phosphoprotein analysis.
High-content imaging systems are being coupled with western blot workflows for enhanced data visualization. These integrated platforms allow researchers to correlate protein expression with cellular localization patterns. The western blot application becomes more powerful when combined with advanced imaging technologies.
Microfluidic devices are miniaturizing western blot procedures while maintaining analytical performance. These systems reduce sample volumes and reagent consumption significantly. Researchers benefit from faster processing times and reduced experimental costs.
Artificial intelligence algorithms are being integrated into data analysis workflows. These systems can identify patterns in phosphorylation data that might escape human observation. Machine learning applications enhance both data interpretation and experimental design optimization.
Potential for Personalized Medicine Applications
Phosphorylation profiling is emerging as a critical component in precision therapeutic approaches. Clinical researchers are using phosphoprotein signatures to predict treatment responses. This application transforms western blot from a research tool into a diagnostic platform.
Biomarker development programs increasingly rely on phosphorylation analysis for patient stratification. These markers help clinicians select optimal treatment protocols for individual patients. The technology enables more targeted and effective therapeutic interventions.
Point-of-care testing devices incorporating western blot principles are under development. These portable systems could bring phosphoprotein analysis directly to clinical settings. Rapid diagnostic capabilities would revolutionize patient care and treatment monitoring.
Companion diagnostic applications are expanding the clinical utility of phosphorylation analysis. These tests help identify patients who will benefit from specific targeted therapies. Personalized treatment protocols based on phosphoprotein profiles show promising clinical outcomes.
| Technology Advancement | Current Capability | Future Potential | Clinical Impact |
|---|---|---|---|
| Fluorescent Detection | Multiplexed protein analysis | Real-time monitoring | Enhanced diagnostic accuracy |
| AI Integration | Pattern recognition | Predictive modeling | Personalized therapy selection |
| Microfluidic Systems | Miniaturized assays | Point-of-care testing | Rapid clinical decisions |
| Mass Spec Coupling | Molecular confirmation | Comprehensive profiling | Precision biomarker discovery |
The future of phosphorylation research lies in the seamless integration of multiple analytical approaches. We anticipate continued improvements in antibody specificity for phosphorylation detection. These advances will enable more precise and reliable experimental outcomes.
Investment in emerging technologies will drive the next wave of scientific discoveries. Research institutions and biotechnology companies are positioning themselves for these technological transitions. The convergence of innovation and application promises exciting developments in protein analysis methodologies.
Conclusion: The Impact of Western Blotting on Protein Research
Western blot methodology continues to serve as the cornerstone technique for phosphoprotein detection in modern laboratories. This established approach provides researchers with reliable, quantitative data essential for understanding cellular mechanisms and disease pathways.
Summary of Key Findings
Our exploration reveals that Western blot remains the most widely adopted method for assessing protein phosphorylation states. The technique’s specificity, combined with phospho-specific antibodies, delivers precise results that other methods struggle to match. Proper sample preparation, optimized protocols, and careful troubleshooting ensure consistent outcomes across diverse research applications.
The Role of Western Blotting in Advancing Biotechnology
Signal transduction analysis through Western blot techniques drives significant breakthroughs in biotechnology and pharmaceutical development. Cancer research, neurological studies, and drug discovery programs rely heavily on this methodology to validate therapeutic targets and monitor treatment responses. The technique’s adaptability allows researchers to investigate complex signaling networks with confidence.
Final Thoughts on Phosphorylation Studies
As research demands evolve, Western blot technology adapts to meet increasing sensitivity and throughput requirements. Integration with complementary techniques enhances analytical capabilities while preserving the fundamental advantages that make this approach indispensable. We anticipate continued innovation in detection technologies will further strengthen Western blot’s position in phosphoprotein research, ensuring its relevance for future scientific discoveries.
References and further readings:
1.Hunter T. A Journey from Phosphotyrosine to Phosphohistidine and Beyond. Mol Cell. 2022;82(11):2111-2124. doi:10.1016/j.molcel.2022.05.007. PMID:356540432.Köhler N, Miri N, Dittrich A. Quantification of Total and Phosphorylated STAT3 by Calibrated Western Blotting. STAR Protoc. 2023;4(3):102508. doi:10.1016/j.xpro.2023.102508. PMID:37669163
3.Kapuy O, Barik D, Domingo Sananes MR, Tyson JJ, Novák B. Bistability by Multiple Phosphorylation of Regulatory Proteins. Prog Biophys Mol Biol. 2009;99(1-2):21-36. doi:10.1016/j.pbiomolbio.2009.06.004. PMID:19523976
4.Tan CSH, Jørgensen C, Linding R. Roles of “Junk Phosphorylation” in Modulating Biomolecular Association of Phosphorylated Proteins? Cell Cycle. 2010;9(7):1277-1282. doi:10.4161/cc.9.7.11066. PMID:20234177
5.McGarvey MJ, Leader DP. The Ribosomal Proteins Phosphorylated in Vitro by Protein Kinase Activities from Krebs II Ascites Cells. Biosci Rep. 1983;3(7):605-614. doi:10.1007/BF01172872. PMID:6578854
FAQ
What makes western blot the gold standard for phosphorylated protein detection?
Western blotting provides unmatched specificity and quantitative capabilities for phosphoprotein detection methods. We utilize this technique because it offers precise molecular weight determination, excellent signal-to-noise ratios, and compatibility with various sample types. The method allows for direct visualization of protein phosphorylation states while maintaining high reproducibility across experiments.
How do phospho-specific antibodies differ from regular protein antibodies?
Phospho-specific antibodies demonstrate antibody specificity for phosphorylation by recognizing only the phosphorylated form of target proteins. We design these antibodies to bind specifically to phosphorylated amino acid residues, typically serine, threonine, or tyrosine. This specificity enables precise phosphorylation site analysis and eliminates interference from non-phosphorylated protein forms.
What are the essential requirements for successful SDS-PAGE phosphoproteins analysis?
Successful SDS-PAGE phosphoproteins analysis requires careful sample preparation with phosphatase inhibitors, optimized gel electrophoresis conditions, and proper transfer protocols. We recommend using gradient gels for optimal protein separation, maintaining cold temperatures during electrophoresis, and employing appropriate blocking strategies to minimize background noise while preserving phosphorylation-dependent signals.
How can I enhance sensitivity when performing immunoblotting phosphoproteins?
We enhance sensitivity through protein loading optimization, enhanced detection substrates, and signal amplification methods. Key strategies include using high-quality phospho-specific antibodies, optimizing antibody dilutions, selecting appropriate detection substrates, and implementing proper incubation conditions. Additionally, we recommend using sensitive imaging systems and optimizing exposure times for maximum signal detection.
What control experiments are crucial for validating phosphorylation-specific signals?
Essential control experiments include phosphatase treatment protocols that validate antibody specificity and confirm phosphorylation-dependent signals. We recommend treating samples with lambda phosphatase or alkaline phosphatase to remove phosphate groups, demonstrating signal loss in treated samples. Additional controls include loading controls, negative controls without primary antibody, and positive controls with known phosphorylated proteins.
How does western blot compare to mass spectrometry for protein kinase activity analysis?
Western blotting provides quantitative analysis of specific phosphorylation sites with high sensitivity, while mass spectrometry offers comprehensive post-translational modifications identification and stoichiometry determination. We utilize western blotting for targeted analysis of known phosphorylation sites and protein kinase activity monitoring, while mass spectrometry complements these studies with site-specific identification and phosphoproteomics profiling.
What are the most common troubleshooting issues in phosphoprotein western blotting?
Common issues include background noise, non-specific binding, and inconsistent band visualization. We address these problems through optimized blocking strategies, proper antibody dilutions, and systematic analysis of experimental variables. Solutions include adjusting blocking reagent selection, optimizing antibody concentrations, improving sample preparation protocols, and validating antibody performance through appropriate controls.
How do I select the appropriate detection method for my protein phosphorylation assay?
Selection depends on research objectives, sample limitations, and required data outputs. We recommend western blotting for targeted signal transduction analysis with high specificity and quantitative capabilities. Consider ELISA for high-throughput screening, mass spectrometry for comprehensive phosphoproteomics, and western blotting for detailed mechanistic studies requiring precise molecular weight determination and signal quantification.
What sample preparation considerations are critical for maintaining phosphorylation states?
Critical considerations include immediate phosphatase inhibitor addition, proper lysis buffer composition, and controlled storage conditions. We recommend using cocktails of phosphatase inhibitors, maintaining samples on ice during processing, and avoiding freeze-thaw cycles that can compromise phosphorylation integrity. Proper protein extraction protocols and rapid processing minimize phosphatase activity and preserve phosphorylation states.
How can western blot analysis contribute to drug development and pharmacodynamics studies?
Western blot analysis provides critical insights into drug mechanism-of-action through protein kinase activity monitoring and phosphorylation status determination. We utilize this technique for time-course studies, dose-response analyses, and biomarker identification in drug development. The method enables precise evaluation of therapeutic targets, pathway modulation, and pharmacodynamic responses in both preclinical and clinical research applications.
Leo Bios
Hello, I’m Leo Bios. As an assistant lecturer, I teach cellular and
molecular biology to undergraduates at a regional US Midwest university. I started as a research tech in
a biotech startup over a decade ago, working on molecular diagnostic tools. This practical experience
fuels my teaching and writing, keeping me engaged in biology’s evolution.




Leave a Comment
Your email address will not be published. Required fields are marked *