The time it takes to run SDS-PAGE changes a lot depending on your method and lab needs. Old methods take at least two hours for the best protein separation. But, new fast methods can do it in just ten minutes.
Lab efficiency is key to research success. New heated buffer systems work at 70-75°C. They make ultra-rapid protein separation possible without losing data quality. This is a big step up from old room-temperature methods.
Now, you can go from starting a sample to detecting it in just one hour. This is a big change from the old two-day process many labs still use. Your choice between fast and slow methods depends on your research needs and what equipment you have.
Many things affect gel running duration. These include voltage, gel type, and the protein you’re working with. Knowing these helps you make your protocols more efficient while keeping science standards high.
Key Takeaways
- Traditional SDS-PAGE protocols require minimum 2 hours for complete protein separation
- Rapid techniques using heated buffers achieve separation in 10 minutes at 70-75°C
- Complete workflow can be reduced from 2 days to 1 hour with optimized methods
- Voltage settings and gel composition significantly impact running duration
- Protocol selection should balance speed requirements with resolution quality
- Modern equipment enables substantial time savings without compromising results
Understanding Western Blotting and Its Importance
Western blotting is the top method for finding proteins in research. It lets scientists accurately spot and measure specific proteins in complex samples. This method combines several steps to give reliable results that lead to new discoveries in many fields.
Today’s labs use Western blotting for its high accuracy and sensitivity. It gives detailed info on protein levels, sizes, and changes after proteins are made. Protein transfer time optimization is key to getting consistent and reliable results.
What is Western Blotting?
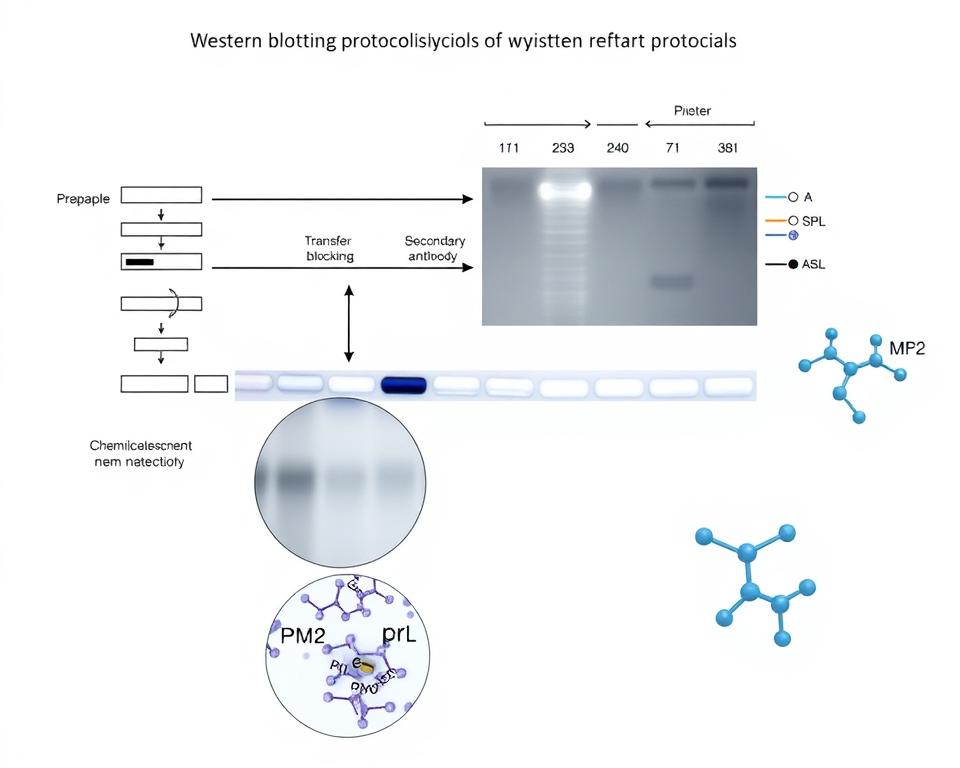
Western blotting is a detailed method that separates, moves, and finds proteins. It starts with sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) to sort proteins by size. Then, proteins move from the gel to a solid membrane.
Next, proteins are found using specific antibodies. These antibodies stick to the target proteins very well. Then, secondary antibodies with enzymes or colors show where the proteins are.
Getting good results from Western blotting needs careful attention to each step. Gel electrophoresis protocols must be fine-tuned for the proteins being studied. Things like temperature, buffer mix, and timing all affect the outcome.
Applications of Western Blot in Research
Cancer research uses Western blotting a lot to study cancer genes and proteins. Scientists compare protein levels in cancer cells to normal cells. This helps find new treatments and markers for cancer.
In neuroscience, Western blotting helps study brain proteins and receptors. It looks at protein changes in brain disorders and development. Protein transfer time optimization is very important here because of the delicate brain tissue.
Drug development also relies on Western blotting to see how drugs affect proteins. Companies use it to check how drugs work and if they are safe. This method gives important data for drug approval and clinical trials.
Key Components of Western Blotting
The Western blotting system has many important parts that work together well. Each part is crucial for the success and trustworthiness of the analysis. Knowing these parts helps researchers improve their work.
| Component | Function | Critical Parameters | Optimization Impact |
|---|---|---|---|
| Electrophoresis System | Protein separation by size | Voltage, gel concentration, buffer pH | Resolution and band sharpness |
| Transfer Apparatus | Protein immobilization on membrane | Current, time, membrane type | Transfer efficiency and protein retention |
| Detection System | Signal visualization and quantification | Antibody concentration, incubation time | Sensitivity and background reduction |
| Imaging Equipment | Data capture and analysis | Exposure time, resolution settings | Quantitative accuracy and reproducibility |
Preparing gels needs the right acrylamide mix for the protein sizes. More acrylamide is better for smaller proteins. Less acrylamide works better for bigger proteins.
Choosing the right membrane is key for protein binding and detection. Nitrocellulose membranes work well for most proteins. PVDF membranes are durable and can be reused several times.
Buffer systems keep the right pH and salt levels during the process. Tris-glycine buffers are the usual choice. But, bis-tris and tricine systems are better for certain protein sizes and gel electrophoresis protocols.
The Steps Involved in Western Blotting
Western blotting has four key stages that affect your results. Each step has its own Western blotting timing factors to consider. This ensures you get the best protein analysis.
Western blotting is a step-by-step process. Delays in one step can impact the whole process. It’s important to plan each step carefully to keep your samples intact and get reliable results.
Sample Preparation
Starting with the right sample preparation is crucial. Choose the right lysis buffers for your proteins. RIPA buffer is good for membrane-bound proteins, while M-PER buffer is gentler.
Add protease and phosphatase inhibitors right away to stop protein breakdown. This step takes 15-30 minutes, depending on your sample and method. Keeping samples on ice helps keep proteins stable.
Gel Electrophoresis
Gel electrophoresis separates proteins by size. The gel’s concentration affects how well proteins are separated. Higher concentration gels are better for smaller proteins but take longer.
The voltage you use also affects how long it takes. Using 100-150V is standard to avoid overheating. But, higher voltage can make the process faster but less precise.
Transfer to Membrane
Transferring proteins from gel to membrane is the next step. Wet transfer methods are best but take 1-2 hours. Semi-dry systems are faster, taking 30-60 minutes.
The type of membrane you use also matters. PVDF membranes need methanol but bind proteins well. Nitrocellulose membranes are easier to handle and transfer faster.
Immunoblotting
Immunoblotting involves several steps to detect proteins. Blocking non-specific sites takes 1 hour at room temperature or overnight at 4°C. The time for primary antibody incubation varies based on the antibody’s strength and amount.
Secondary antibody application takes 1-2 hours for the best results. The final step is substrate development, which depends on the detection method and desired intensity.
| Western Blot Step | Typical Duration | Key Timing Factors | Optimization Strategies |
|---|---|---|---|
| Sample Preparation | 15-30 minutes | Lysis buffer type, sample volume | Pre-chill buffers, use efficient lysis methods |
| Gel Electrophoresis | 45-90 minutes | Gel concentration, voltage, protein size | Optimize voltage settings, use precast gels |
| Membrane Transfer | 30-120 minutes | Transfer method, membrane type | Semi-dry systems, optimized buffer conditions |
| Immunoblotting | 4-24 hours | Antibody concentrations, incubation conditions | Room temperature incubations, optimized dilutions |
Every step in Western blotting adds to the total time. Electrophoresis is the most variable part, depending on your needs and strategies. Knowing these timing considerations helps plan your experiments better and get consistent results.
Factors Influencing Electrophoresis Time
The time it takes for electrophoresis depends on several key factors. To get the best results, researchers need to understand how these factors work together. This knowledge helps in achieving optimal Western blot conditions.
Each factor has its own role in how fast and well proteins separate. By understanding these relationships, researchers can fine-tune their protocols to meet their specific needs.
Gel Concentration
The concentration of acrylamide in the gel is crucial for protein movement. Higher concentrations mean smaller pores, which slow proteins down but improve resolution for similar-sized proteins.
Gradient gels with a range of 4-20% are versatile. They can handle a wide range of protein sizes in one run, but they take longer than uniform gels.
For proteins between 10-100 kDa, 12-15% gels are best. They offer good resolution and reasonable run times, making them ideal for specific molecular weight ranges.
Size of Protein
The size of a protein affects how fast it moves through the gel. Larger proteins face more resistance and move slower than smaller ones.
Small proteins under 20 kDa move quickly and may need higher gel concentrations for clear separation. On the other hand, proteins over 150 kDa move slowly and benefit from lower concentration gels to reduce resistance.
Knowing the size of your target protein helps predict how long it will take to separate. This information is crucial for planning your experiment and managing resources.
Voltage Settings
Voltage is what drives protein migration and affects both speed and quality. Higher voltages make separation faster but can also increase heat, which can distort bands.
For standard protocols, we suggest voltages between 100-150V. This range provides good separation in 1-2 hours, balancing speed with heat to keep proteins intact.
For faster separation, higher voltages with temperature control can be used. These methods can cut separation time to 30-45 minutes while keeping quality high through cooling.
| Gel Concentration | Optimal Protein Size Range | Typical Voltage | Expected Run Time |
|---|---|---|---|
| 4-20% Gradient | 5-250 kDa | 120-150V | 90-120 minutes |
| 12% Uniform | 20-100 kDa | 100-130V | 60-90 minutes |
| 15% Uniform | 10-70 kDa | 100-120V | 75-105 minutes |
| 8% Uniform | 50-200 kDa | 80-110V | 90-120 minutes |
By balancing these three factors, researchers can create optimal Western blot conditions for their specific needs. Consider your protein targets, available time, and required resolution when choosing electrophoresis parameters for the best results.
Typical Timeframes for Each Step
Knowing the exact time for each Western blot step helps plan better. We give detailed timing guides to help you use your time wisely. These times are useful for both new and experienced researchers.
Western blot has different steps that take different amounts of time. The whole process can take 6 to 12 hours. This depends on the method and the proteins you’re working with. It’s important to plan well, especially when you have many experiments or tight deadlines.
Sample Preparation Duration
Getting your samples ready takes 30 to 60 minutes. This includes breaking down cells, removing debris, and measuring protein amounts. You might need more time for special treatments.
Denaturing steps need extra time. For example, heating samples at 70°C for 10 minutes is needed for some conditions. Molecular weight transfer considerations start here, as proper denaturation is key for good separation.
Harder samples might take up to 90 minutes to prepare. This includes tissues, membrane proteins, or hard-to-break cells. It’s smart to prepare buffers and set up equipment early to avoid delays.
Electrophoresis Time
Traditional electrophoresis takes 90 to 120 minutes at 100-150V. This time is needed for good separation in mini-gels. Higher voltages can be faster but might not be as good.
Rapid systems can separate proteins in just 10 minutes. They use special buffers and gels. But, traditional methods often give better results for complex mixtures.
The gel’s concentration affects how long electrophoresis takes. Thicker gels are better for big proteins, while thinner ones are better for small ones. You need to find a balance between time and the quality of separation.
Transfer Time
Wet transfer methods take 2 to 4 hours at 70V. Overnight transfers at 30V work well but take longer. These methods are great for transferring big proteins.
Rapid semi-dry transfer systems can transfer proteins in 7 minutes. They use special buffers and high currents. Molecular weight transfer considerations are key when choosing transfer conditions.
Transfer efficiency depends on protein size and membrane type. Big proteins need longer times or special conditions. It’s important to adjust transfer settings for your specific proteins and needs.
Antibody incubation periods come after transfer. Primary antibodies need 1-2 hours, and secondary ones need 1 hour. Faster methods can cut these times to 10 minutes each.
Total antibody incubation periods can be overnight for better sensitivity. This is good for finding low-abundance proteins but takes longer. You need to balance sensitivity needs with time limits when planning.
Comparing Electrophoresis Times Across Methods
Different electrophoresis methods have different times and ways to separate proteins for Western blots. We compare these to help you pick the best method for your research. Knowing the time differences helps plan your work better.
Each method has its own benefits like speed, resolution, and how many samples it can handle. Your choice depends on how fast you need the results and how well you need them separated.
SDS-PAGE vs. Native Gel Electrophoresis
SDS-PAGE is the usual method for Western blots. It takes longer because proteins are unfolded and have the same charge. It usually takes 90 to 120 minutes to separate all proteins.
Native gel electrophoresis is faster, taking 60 to 90 minutes. Proteins keep their natural shape and charge. But, the separation is different from SDS-PAGE.
Native methods keep protein complexes and activities. But, they might not separate proteins as well as SDS-PAGE. The Western blot application decides which method is best for your research.
High-Throughput Techniques
High-throughput systems use automated platforms and special buffers. They make analysis much faster. Some can do a whole Western blot in under three hours.
These systems do everything automatically. This means less time spent on each step. High-throughput methods are great for labs that need to process many samples every day.
Investing in these systems means you can handle more samples. You get consistent results and save time in the lab.
Capillary Electrophoresis
Capillary electrophoresis is the fastest method. It can separate proteins in 10 to 30 minutes. This is because of the narrow capillaries and high voltage.
But, it can only handle a few samples at a time. It’s best for quick analysis of one sample. It’s fast but can’t handle many samples.
It’s perfect for when you need results fast. But, think if you need to process many samples before choosing.
| Electrophoresis Method | Separation Time | Sample Capacity | Resolution Quality | Best Application |
|---|---|---|---|---|
| SDS-PAGE | 90-120 minutes | High | Excellent | Standard Western blotting |
| Native PAGE | 60-90 minutes | High | Good | Protein complex analysis |
| High-Throughput | 45-75 minutes | Very High | Excellent | Multiple sample processing |
| Capillary | 10-30 minutes | Low | Very Good | Rapid single analysis |
Choosing a method affects both Western blot electrophoresis time and your results. Think about your lab’s needs before picking a method. Consider how many samples you have, how fast you need results, and how detailed you need them to be.
Optimizing Electrophoresis Efficiency
To improve protein transfer time optimization, focus on a few key areas. These help speed up your work without losing quality. By doing so, you can make your lab more efficient and precise.
Modern labs can save a lot of time by tweaking their protocols. It’s all about understanding how changes in separation affect protein quality.

High – precision Electrolysis Reactor
Tips for Reducing Electrophoresis Time
One fast way to separate proteins is by using a heated running buffer at 70-75°C. This method cuts down your time to just 10 minutes without losing quality.
When using heated buffers, watch the temperature closely. Make sure you have good ventilation and the right equipment to avoid overheating.
Here are more ways to save time:
- Start with a low voltage and increase it to 200V for quicker migration
- Use more acrylamide for small proteins
- Warm up the gel apparatus before adding samples
- Adjust the sample volume to speed up loading
When to Consider Precast Gels
Precast gels are great for everyday use where consistency is key. They save 2-3 hours of prep time and ensure consistent results.
Choose precast gels when you need:
- Consistent results across many experiments
- Longer shelf life for occasional use
- Less time spent on prep
- Standardized protocols for regulatory needs
While hand-cast gels might be cheaper for big labs, the time and consistency of precast gels are often worth the extra cost.
Utilizing Different Buffers
The buffer you choose greatly affects how well proteins separate in gel electrophoresis protocols. Bis-Tris gels are faster and keep proteins stable better than Tris-glycine.
Tricine buffers are better for small proteins but take longer. Make sure the buffer works well with your proteins and detection methods.
Here are some buffer tips:
- Bis-Tris systems: Fast and keeps pH stable
- Tris-glycine buffers: Good for most protein sizes
- Tricine buffers: Best for proteins under 30 kDa
Try different buffers to find the best mix of speed and quality for your needs. This ensures your work is efficient and accurate.
Assessing Protein Size and Its Impact
The connection between protein traits and Western blot time is key. Knowing how size affects separation is crucial. Before setting up your run, check your protein’s size.
Protein size affects how long it takes to separate them. Bigger proteins move slower through the gel. This means longer times for high-weight proteins.
Size Determination in Western Blot
Molecular weight standards are vital for identifying proteins. They help place your proteins on the gel. These markers create a guide for accurate size.
Choose markers that cover your protein’s size range. This ensures clear separation and identification. Prestained markers offer real-time visualization during runs.
The gel’s concentration should match your protein’s size. More acrylamide is needed for smaller proteins. Less is better for larger ones.
| Protein Size Range (kDa) | Recommended Gel Concentration (%) | Typical Electrophoresis Time (hours) | Optimal Voltage (V) |
|---|---|---|---|
| 10-50 | 12-15 | 1.0-1.5 | 120-150 |
| 50-100 | 8-12 | 1.5-2.0 | 100-120 |
| 100-200 | 6-8 | 2.0-2.5 | 80-100 |
| 200+ | 4-6 | 2.5-3.0 | 60-80 |
Implications for Electrophoresis Time
Timing is everything for proteins over 100 kDa. They need more time to separate well. Inadequate time leads to poor results.
Proteins move through gels in a pattern based on size. Smaller ones go faster, needing less time. Proteins under 20 kDa usually separate in 60-90 minutes.
Changes after protein synthesis can affect size. Glycosylated proteins, for example, move slower. Adjust your timing for these changes.
Gradient gels are versatile for unknown proteins or complex samples. They work well across a wide range of sizes. But, they might need longer times for best results.
Keeping the temperature steady is key for long runs. Heat can mess with protein movement and gel quality. Use coolers for runs over two hours.
Common Mistakes That Extend Western Blot Time
Identifying and avoiding common Western blot mistakes can save a lot of time. Most delays come from three main areas that researchers often miss. These mistakes can turn quick experiments into long ones.
Knowing these common errors helps keep your Western blot efficient. It also boosts your lab’s productivity. Understanding these issues is key to maintaining optimal Western blot conditions.
Inadequate Sample Preparation
Not preparing samples well is the top reason for longer Western blot times. Incomplete protein denaturation happens when samples aren’t heated enough or for long enough.
Make sure to heat your samples for exactly 10 minutes. Check that your lysate is clear before moving on to the next step.
Bad lysis techniques also cause problems. Not spinning samples enough can clog gel wells and mess up protein movement.
Misjudging Gel Concentration
Choosing the wrong acrylamide percentage can ruin your electrophoretic separation efficiency. Incorrect gel selection can make it hard to see target proteins or take too long.
Always check protein size charts before making your gel. Use gradient gels if you’re not sure about protein sizes.
Using old acrylamide or storing reagents wrong can mess up gel density. This makes separation poor and takes longer.
Incorrect Voltage Settings
Choosing the wrong voltage can make your run too long or ruin band sharpness. Optimal voltage settings are key to balancing speed and quality.
Stick to 100-150V for standard protocols. Higher voltages can damage proteins and lower data quality.
Make sure your buffers and equipment are at room temperature before starting. This avoids thermal shock and keeps results consistent.
| Common Mistake | Time Impact | Solution | Prevention Strategy |
|---|---|---|---|
| Insufficient protein denaturation | +30-60 minutes | Heat samples at 70°C for 10 minutes | Verify clear lysate appearance |
| Wrong gel concentration | Complete protocol repetition | Consult protein size charts | Use gradient gels for unknown sizes |
| Excessive voltage settings | +45-90 minutes | Maintain 100-150V range | Monitor buffer temperature |
| Expired reagents | Variable delays | Check expiration dates | Implement inventory rotation |
Case Studies: Electrophoresis Time in Practice
Different labs use various timing strategies for electrophoresis. We look at how labs in real-world settings have optimized their timing. These examples show how molecular weight transfer considerations and workflow needs shape their protocols.
Labs have different priorities and constraints. Research labs focus on quality, while clinical labs need fast results for patient care.
Academic Research Applications
University labs often choose quality over speed. Students use overnight runs at 50-60 volts for top-notch protein results.
At Stanford University, a cancer research lab uses 3-hour electrophoresis for tumor protein analysis. They focus on molecular weight transfer considerations during blotting. This method separates proteins well, from 15 kDa to 250 kDa.
Academic labs sometimes analyze many proteins at once. They can use longer protocols because of flexible schedules. But, tight thesis deadlines might push them to quicker methods.
- Extended electrophoresis times of 2-3 hours for broad molecular weight ranges
- Overnight runs at low voltages for maximum resolution
- Flexible scheduling accommodating longer protocols
- Quality prioritized over speed for publication requirements
Clinical Laboratories Perspective
Clinical labs need fast, consistent results for patient care. Mayo Clinic’s immunology department uses automated Western blots. These systems take 3-4 hours, much faster than manual methods.
Clinical labs require strict protocols and minimal changes. Antibody incubation periods are standardized for consistent results. They often prefer precast gels and set conditions over quick methods.
Regulations limit changes in clinical labs. Any new timing methods must be thoroughly tested before use. This makes it hard to adopt new, faster techniques, even if they work well in research.
| Laboratory Type | Typical Electrophoresis Time | Primary Priority | Flexibility Level |
|---|---|---|---|
| Academic Research | 2-3 hours | Data Quality | High |
| Clinical Diagnostic | 45-90 minutes | Speed & Consistency | Low |
| Pharmaceutical R&D | 1-2 hours | Throughput | Medium |
| Contract Testing | 60-120 minutes | Cost Efficiency | Medium |
Clinical labs are moving to automated systems. These systems help keep antibody incubation periods consistent and cut down analysis time. They meet rules and make workflows more efficient.
Optimizing timing is about finding the right balance for each lab. Labs need to consider their specific needs and follow rules.
Innovations in Western Blotting Techniques
Modern technology is changing Western blot electrophoresis time with new gel formulas and automated systems. These changes help labs work faster and more accurately. They make protein analysis more efficient.
New ideas focus on better gel chemistry and automated systems. These improvements cut down the time needed for analysis. You get faster and more reliable results.
Advances in Gel Technology
New polyacrylamide formulas let proteins move faster without losing quality. Modern gels finish electrophoresis in 15-20 minutes. They keep the quality high while making SDS-PAGE running duration shorter.
Improved pre-cast gels have better gradients and stability. This means less time preparing and more consistent results. Novel buffer compositions also speed up protein movement.
Capillary-based systems are big steps forward in protein separation. They separate proteins in minutes, not hours. High-voltage gradients through narrow channels make analysis quick without losing detail.
| Technology Type | Electrophoresis Time | Sample Volume | Throughput Capacity |
|---|---|---|---|
| Traditional SDS-PAGE | 60-90 minutes | 20-50 μL | 10-20 samples |
| Fast-Cast Gels | 15-20 minutes | 15-30 μL | 15-25 samples |
| Capillary Systems | 5-10 minutes | 1-5 μL | 96-384 samples |
| Microfluidic Platforms | 3-8 minutes | 0.1-1 μL | 100+ samples |
Integration with Automated Systems
Robotic systems take over the electrophoresis process. They handle everything from loading samples to starting transfers. This makes Western blot electrophoresis time consistent.
These systems cut down the time needed for Western blots from days to hours. They also reduce mistakes. Integrated detection systems let you watch the process in real-time.
Microfluidic Western blot platforms are new and fast. They do the whole analysis in 60-90 minutes. They use tiny amounts of sample and have built-in detection.
When looking at these new technologies, think about how much you need to do and your budget. SDS-PAGE running duration improvements should fit your research needs. We suggest looking at how automation can help your lab work better.
Future Directions for Electrophoresis in Western Blotting
New technologies are coming that will make Western blotting faster and more precise. We’re seeing big changes in how we do electrophoresis. These changes will make labs work better and faster.
People want to detect proteins quicker and more reliably. We’ll see better hardware and software soon. These will be easy to use but still very accurate.
Trends in Sample Processing
Miniaturization is changing how we process samples. Now, we can analyze proteins from just one cell or a tiny amount. This makes things much faster than before.
Systems that do everything automatically are becoming common. They cut down on mistakes and make results more consistent. Digital Western blot platforms use tiny cartridges for better results.
New ways to quickly break down samples are being developed. These systems make the whole process smoother. They’re getting closer to being ready for use in clinics with little training needed.
New gel electrophoresis methods work at room temperature. They can give results in about an hour. These systems are getting even better.
Potential Time-Saving Approaches
Using artificial intelligence can make protocols better automatically. Systems that watch electrophoresis in real-time adjust settings as needed. They learn from how proteins move.
Multiplexed detection lets us look at many proteins at once without taking longer. New buffers help separate proteins fast at room temperature. This makes everything faster.
The table below shows how different technologies will make Western blotting faster:
| Technology Approach | Current Processing Time | Projected Future Time | Key Advantage | Implementation Timeline |
|---|---|---|---|---|
| Digital Western Platforms | 4-6 hours | 60-90 minutes | Automated workflow | Currently available |
| Microfluidic Systems | 3-4 hours | 30-45 minutes | Minimal sample volume | 2-3 years |
| AI-Optimized Protocols | Variable timing | Optimized automatically | Adaptive optimization | 3-5 years |
| Room-Temperature Buffers | Standard timing | 25% time reduction | No cooling required | 1-2 years |
Adding mass spectrometry to Western blotting makes analysis even better. These systems combine different techniques for detailed protein analysis. Point-of-care applications will keep giving top-notch results thanks to new tech.
We expect to see more standardization in detecting many proteins at once. Watching the process in real-time will become key for better results. Labs will keep getting better at working with different methods.
Improving Western blotting means keeping quality high but using less space and equipment. New tech will make it easier for everyone to use without losing scientific quality. This will help Western blotting grow in both clinics and research.
Summary and Key Takeaways
Understanding Western blotting timing factors helps you make better choices. We’ve seen how different methods and needs affect electrophoresis time.
Timing Considerations for Protocol Selection
Your electrophoresis time depends on several factors. Traditional SDS-PAGE takes 60-90 minutes. But, rapid methods can finish in 10-15 minutes with heated buffers.
The gel electrophoresis step usually takes about 1 hour with the right voltage.
Protein size, gel concentration, and voltage all affect how fast proteins separate. Smaller proteins move faster in higher concentration gels. Larger proteins need lower concentration gels for best results.
Workflow Enhancement Strategies
To get the best Western blot results, balance speed and quality. Choose the right equipment and methods to save time. Precast gels save prep time, automated systems reduce manual work, and fast transfer methods speed up membrane processing.
Consider your research needs and available time. Quick methods are great for high-throughput studies. But, detailed protein analysis might need longer separations for better results and reproducibility.
References and further readings:
1.Mahmood T, Yang PC. Western blot: technique, theory, and trouble shooting. North Am J Med Sci. 2012;4(9):429-434.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3456489/2.Kurien BT, Scofield RH. Western blotting. Methods. 2006;38(4):283-293.
https://www.sciencedirect.com/science/article/abs/pii/S1046202306000065?via%3Dihub3.Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and applications. Proc Natl Acad Sci USA. 1979;76(9):4350-4354.
https://www.pnas.org/doi/abs/10.1073/pnas.76.9.43504.Burnette WN. “Western blotting”: electrophoretic transfer of proteins from sodium dodecyl sulfate–polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981;112(2):195-203.
https://www.sciencedirect.com/science/article/abs/pii/0003269781902815?via%3Dihub
FAQ
How long does electrophoresis typically take for Western blot analysis?
Electrophoresis for Western blot analysis can take anywhere from 10 minutes to 3 hours. This depends on the gel concentration, protein size, and voltage. Rapid techniques can separate proteins in just 10 minutes with heated buffers.
What factors most significantly influence Western blot electrophoresis time?
Several factors affect electrophoresis time. These include gel concentration, protein size, and voltage. Balancing these factors is key to achieving good results.
Can I reduce electrophoresis time without compromising protein separation quality?
Yes, you can speed up electrophoresis by heating Laemmli buffer to 70-75°C. This method requires careful temperature control. Precast gels also save time and ensure consistency.
How does protein size affect electrophoresis timing in Western blot protocols?
Larger proteins take longer to separate, up to 3 hours. Smaller proteins can be separated in 60-90 minutes. The time needed increases with protein size.
What are the complete timeframes for all Western blot steps?
Preparing samples takes 30-60 minutes. Electrophoresis time varies from 10 minutes to 3 hours. Transfer times range from 7 minutes to overnight. Antibody incubation takes 1-2 hours for primary and 1 hour for secondary antibodies. Total time ranges from 1 hour to 2 days.
How do SDS-PAGE and native gel electrophoresis timing compare?
SDS-PAGE takes 90-120 minutes due to protein unfolding. Native gel electrophoresis is faster, 60-90 minutes, as proteins keep their native shape. However, the separation patterns are different.
What voltage settings provide optimal electrophoresis timing?
Use 100-150V for standard protocols, which takes 1-2 hours. Higher voltages can distort bands and should be avoided.
When should I consider using precast gels for Western blot electrophoresis?
Use precast gels for routine applications to save time. They offer consistency but may be more expensive than hand-cast gels for high-volume labs.
What are the most common mistakes that extend Western blot electrophoresis time?
Common mistakes include poor sample preparation and incorrect voltage settings. Ensure complete protein solubilization and choose the right gel concentration.
How do rapid Western blot systems compare to traditional methods in terms of timing?
Rapid systems achieve results in 7 minutes, compared to 2-4 hours for traditional methods. Automated platforms can reduce time from days to hours. Digital systems analyze proteins in 60 minutes.
What buffer systems can help reduce electrophoresis running duration?
Bis-Tris buffer systems are faster and more stable than Tris-glycine. Heated Laemmli buffer can speed up electrophoresis to 10 minutes. Tricine systems offer better resolution for small proteins but take longer.
How does capillary electrophoresis timing compare to traditional gel electrophoresis?
Capillary electrophoresis is the fastest, taking 10-30 minutes. However, it has limited sample capacity, making it less suitable for multi-sample workflows.
What innovations are reducing Western blot electrophoresis time?
New polyacrylamide formulations can separate proteins in 15-20 minutes. Microfluidic platforms analyze proteins in 60-90 minutes. Automated systems and AI optimize protocols in real-time.
How do molecular weight transfer considerations affect electrophoresis timing?
Larger proteins require longer separation times. Gradient gels are versatile for unknown targets, while fixed concentration gels offer better resolution for specific sizes.
What are the optimal Western blot conditions for minimizing total protocol time?
Use heated buffer electrophoresis, rapid transfer systems, precast gels, and optimized antibodies. These can reduce total time to under 3 hours while maintaining quality.
Leo Bios
Hello, I’m Leo Bios. As an assistant lecturer, I teach cellular and
molecular biology to undergraduates at a regional US Midwest university. I started as a research tech in
a biotech startup over a decade ago, working on molecular diagnostic tools. This practical experience
fuels my teaching and writing, keeping me engaged in biology’s evolution.





Leave a Comment
Your email address will not be published. Required fields are marked *